
Exclusivity Engineered: AI-Driven eLearning Solutions for Retail & Luxury
Gain efficiency while maintaining human artistry with a hybrid AI-human approach.


Case Study: Multilingual Retail Marketing
New AI Content Creation Solutions for a Sports and Apparel Giant


Human Expertise Blended With Powerful AI
Lionbridge Aurora AI™ is an AI-first global content platform that increases your multilingual content creation and expands your audience with culturally relevant, hyper-personalized content.

- RESOURCES


No one with any experience in the pharmaceutical industry would dare intimate that the regulatory process is simple.
Find out how to reduce complexity.
The current clinical research landscape is incredibly vast, and the number of trials occurring worldwide grows higher every year. There are currently 284,522 studies in 204 countries worldwide. That scope engenders excitement—more innovation yields more solutions for more diseases in more populations—but it also brings increasing complexity to an already complicated market.
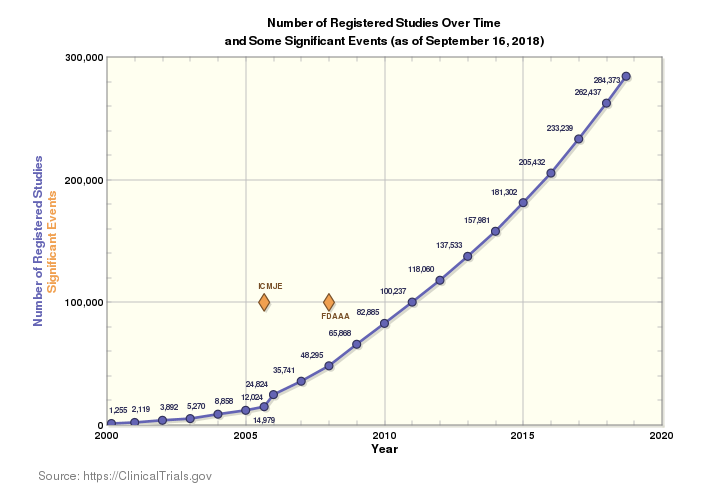
More trials in more countries and more languages mean more rules to follow, more people to consider, and more regulations to track for clinical supply chains. This, coupled with the ever-present push to reduce time to market, lower costs, and eliminate errors in supply and delivery, makes ensuring compliance more complicated than ever.
Must “compliant” mean “complicated?” No. Here are three ways smart companies reduce complexity in the clinical labeling process.
1. Create a certified database for translations and regulatory content.
The process of preparing clinical labels in a way that ensures both accuracy and compliance is largely manual. Multiple contacts must review documentation before its output, numerous vendors have disparate opinions and perspectives, and various parties have different preferences for translations—which can lead pharma companies to “reinvent the wheel” each time they undergo a new clinical labeling initiative.
Certified databases for translations and regulatory content—with consistent and comprehensive phrase libraries—reduce cycle time, standardize content, enable quicker late-stage changes, and reduce risk by maintaining consistency across your global process. Bottom line: Your products get to market faster, cheaper, and with less exposure.
2. Rely on dedicated in-country experts—not your colleagues.
As pharma companies strive to do more with fewer resources, they often try to keep costs down and by managing regulations in-house with “borrowed resources.” But leaning on your colleagues for compliance and completing the clinical labeling process often actually increases both costs and complexity. Why? This “per label” process for regulatory validation and translation yields longer lead times, a higher prevalence of error, and increased difficulty tracking compliance.
Smart organizations know that investing in the right in-country experts with both regulatory and linguistic expertise ensures their labeling is delivered accurately and quickly every time. And by moving away from the “per label” approach and standardizing the process around these experts, they lower their risk profile.
3. Embrace controlled creativity.
Some of the best solutions to the problems that result from increased complexity and a changing regulatory landscape require a little creativity. For example, your organization could minimize rounds of edits and relabeling by getting an intentionally late start on the labeling process, which would also assist in managing storage, logistics and shelf life. Or you could expedite translations with the use of robust phrase libraries and innovative language technology.
Paradoxically, though, in attempting to reduce complexity, these solutions can create complications of their own. Each requires specialist design, development, and management. If you want to solve your problems comprehensively, systematically, and creatively, you’re often best-served by working with a partner who can handle these logistics for you.
At Lionbridge Life Sciences, we’re constantly seeking ways to help our clients overcome the issues that preclude them from getting safe, effective, affordable medicines to the market as fast as possible. Learn more about how you can overcome these issues and what your colleagues are doing to address the ever-increasing complexity of clinical labeling for IMP and IND by watching this recent webinar featuring Lionbridge Life Sciences VP Peter Quigley via DIA or get in contact with us.


